Northern Redbelly × Finescale Dace hybrid
Status of Northern Redbelly × Finescale Dace hybrid (Chrosomus eos × Chrosomus neogaeus) in Montana
 Allison L. Stringer
Allison L. Stringer
U.S. Forest Service
Bozeman, MT
allison.stringer@usda.gov
Shannon Blackburn
Montana Fish, Wildlife & Parks
Billings, MT
Shannon.blackburn@mt.gov
May 2020
DESCRIPTION
The Northern Redbelly × Finescale Dace hybrid Chrosomus eos × Chrosomus neogaeus, (hereafter, hybrid dace) is a small-bodied minnow (family Leuciscidae, syn. Cyprinidae) native to the United States and Canada. Hybrid dace must co-occur with Northern Redbelly Dace or Finescale Dace because they reproduce clonally by gynogenesis (Goddard et al. 1998; Angers and Schlosser 2007; Mee and Taylor 2012). In Montana, hybrid dace only co-occur with Northern Redbelly Dace because Finescale Dace have not been documented in the state (Stringer 2018).
Hybrid dace have physical characteristics that are intermediate between Northern Redbelly Dace and Finescale Dace (New 1962; Schlosser et al. 1998; Mee and Rowe 2010). However, considerable morphological variation exists in both parental species and their hybrid, and most biologists cannot reliably distinguish hybrid dace from their parental species in the field (Goddard et al. 1989). In the lab, hybrid dace are definitively identified using pharyngeal tooth counts, intestinal complexity, genetic testing, or a combination of the three (New 1962). In Montana, Northern Redbelly Dace occur more widely and in higher numbers than their hybrids, and most hybrid dace are probably misidentified as Northern Redbelly Dace in the field. Misidentification has caused some confusion about their statuses and co-occurrence in the past (Stringer 2018).
LIFE HISTORY, BEHAVIOR, AND ECOLOGY
Hybrid dace exhibit a unique life history, as they primarily reproduce through the process of gynogenesis (Goddard et al. 1998; Angers and Schlosser 2007; Mee and Taylor 2012). Therefore, most hybrid dace produce clonal, diploid ova, which can only develop after stimulation by sperm from a male Finescale or Northern Redbelly Dace. Most of the time, the egg does not incorporate any of the sperm’s genetic material and develops into a maternal clone (Goddard et al. 1989). However, sometimes the sperm is fully or partially incorporated into the ovum, resulting in triploid or diploid-triploid mosaic fish (Dawley et al. 1987; Dawley and Goddard 1988; Goddard et al. 1989; Goddard and Dawley 1990; Doeringsfeld et al. 2004; Binet and Angers 2005; Angers and Scholosser 2007). The triploid and mosaic individuals cannot reproduce gynogenetically, instead producing normal haploid ova which contain only the third chromosome incorporated from the parental species. Sperm from either parental species can then fertilize this egg, resulting in a genetically pure individual the present parental species. In Montana, this process results in pure Northern Redbelly Dace that contain Finescale Dace mitochondrial DNA (Goddard et al. 1998), dubbed cybrids (Binet and Angers 2005; Mee and Taylor 2012). Cybrids have completely replaced Northern Redbelly Dace in Montana and other northern tier states (Mee and Taylor 2012).
Hybrid dace do not originate from recent hybridization events, but rather from multiple hybridization events between female Finescale Dace and male Northern Redbelly Dace that took place in the Mississippi glacial refuge during the Pleistocene epoch (< 50,000 year ago; Angers and Schlosser 2007; Mee and Taylor 2012). After proglacial lakes retreated, hybrid dace and their parental species dispersed across the continent. However, Finescale Dace do not currently occur in Montana (Stringer 2018), suggesting that they were extirpated at some point in the past, or that they did not disperse here at all. Therefore, no future hybridization can occur, and the limited number of clonal lineages in Montana will continue to have low genetic diversity.
Few studies have documented the ecology of hybrid dace, but they probably display behavior and a life history intermediate between parental species. In fact, the niche of hybrid dace probably overlaps with both the mostly insectivorous Finescale Dace and the mostly herbivorous Northern Redbelly Dace, resulting in a more “general purpose” phenotype (Schlosser et al. 1998). Hybrid dace may have gained additional physiological flexibility through hybridization. For example, hybrid dace survive longer in oxygen stressed environments, consume a wider variety of food items, and exist in more varied physiochemical environments than either parental species (Schlosser et al. 1998; Mee and Rowe 2010). In certain environments hybrid dace seem to have a competitive advantage over Northern Redbelly Dace (Stasiak 1972; Stasiak 2006), whereas in others they do not appear to occur at all (Stasiak 1972; Felts 2013).
DISTRIBUTION AND HABITAT
The range of hybrid dace overlaps with those of both parental species and includes much of southern Canada and the northern United States, from eastern British Columbia to Nova Scotia and from Montana to Maine (Lee et al. 1980). However, the exact distribution of hybrid dace is uncertain because of the similarity in external appearance to Northern Redbelly Dace. Relic populations seem to occur with Northern Redbelly Dace in Colorado, South Dakota, and Nebraska (Bestgen 1989; Felts 2013). Similar to Northern Redbelly Dace and Finescale Dace, hybrid dace are widely distributed but appear to be rare in the prairie portion of their range (Bestgen 1989; Stasiak 2006; Felts 2013).
Montana contains the southwestern periphery of the continental range of hybrid dace. Historically, hybrid dace probably occurred in most drainages where Northern Redbelly Dace also occurred (i.e., Missouri, Little Missouri, lower Yellowstone, and Saskatchewan River basins; Figure 1). However, the historical distribution of hybrid dace may have been overestimated as hybrids were only collected in 48% of streams with Northern Redbelly Dace in a study of Northern Redbelly Dace distribution (Stringer 2018). As in other locations, unknown accuracy of past field identifications results in considerable uncertainty. Both Northern Redbelly Dace and hybrid dace appear to have been extirpated from the Little Missouri drainage in Montana sometime between 1993 and 2016 (Stringer 2018). Disjunct populations probably occur in the lower Gallatin and Madison River basins with Northern Redbelly Dace; however, we know little about their abundance or wider distribution in these drainages because no targeted surveys have been conducted.
Hybrid dace occur with either Northern Redbelly Dace or Finescale Dace, and therefore occur in the habitat that their parental species occupy. Both Northern Redbelly Dace and Finescale Dace prefer calm waters of beaver ponds, bogs, and clear streams, with Finescale Dace also occurring in larger lakes (Stasiak 1972; Holton and Johnson 1996; Stasiak 2006; Mee and Rowe 2010). Hybrid dace probably have greater habitat tolerances (e.g., greater physiological response to oxygen stress) than Northern Redbelly Dace and Finescale Dace (Schlosser et al. 1998), and might occur in a wider range of habitats if they did not need to co-occur with one of their parental species for reproduction.
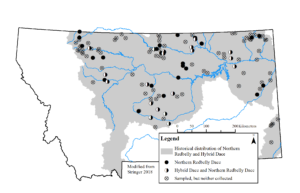
Figure 1. Collections of hybrid dace and Northern Redbelly Dace from their historical distribution in a 2015–2017 study of their current status and distributions in Montana (adapted from Stringer 2018).
CONSERVATION STATUS AND MANAGEMENT
In Montana, the hybrid dace is currently listed as an “S2” species of concern by the MT Chapter AFS because they are at moderate risk of extirpation in the state because of limited and/or declining numbers, range and/or habitat, even though it may be abundant in some areas (Bramblett and Stagliano 2012, MTAFS 2012). Hybrid dace are reported from fewer than 20 populations in the state and relatively little is known about its historical distribution and biology (Figure 1). Hybrid dace and Northern Redbelly Dace populations have declined and are at moderate risk of extirpation in Montana (Stringer 2018). Individuals were collected from only 19 of 40 streams (48%) where Northern Redbelly Dace were also collected (Stringer 2018). Range contraction has probably resulted from the widespread invasion of Northern Pike Esox lucius and non-native trout.. Habitat degradation (e.g., urbanization, grazing, and agriculture practices) has probably also played a role in their decline and has been shown to negatively affect Northern Redbelly Dace and Finescale Dace populations elsewhere (Whittier et al. 1997).
Exclusion or removal of non-native predators affords the best chance of conserving hybrid dace (Stringer 2018). In the Musselshell River, a series of diversion dams probably restrict upstream movement of both native and non-native fishes. Although passage at some diversions is needed to reconnect native Sauger Sander canadensis with historical spawning areas, retention of the Stella and Parrot diversion dams will probably prevent the spread of Northern Pike and other invasive species into the upper Musselshell (M. Ruggles, Montana Fish, Wildlife & Parks, personal communication). Ongoing work will prioritize dam removal and retention in areas that could serve as climate refugia for dace and other native fishes (N. Clancy, unpublished data). Furthermore, managers should carefully consider the consequences of stocking Northern Pike or trout into drainages containing hybrid and Northern Redbelly Dace. Establishing a prairie fish monitoring program would allow for a better understanding of long-term population trends of hybrid dace (and other native prairie fishes) and the potential need for a conservation-hatchery option.
STATUS
REFERENCES
Angers, B., and I. J. Schlosser. 2007. The origin of Phoxinus eos-neogaeus unisexual hybrids. Molecular Ecology 16:4562–4571.
Bestgen, K. R. 1989. Distribution and notes on the biology of Phoxinus eos (Cyprinidae) in Colorado. The Southwestern Naturalist 34:225–231.
Binet, M. C., and B. Angers. 2005. Genetic identification of members of the Phoxinus eos‐neogaeus hybrid complex. Journal of Fish Biology 67(4):1169–1177.
Bramblett, B. and D. Stagliano 2012. Northern Redbelly Dace x Finescale Dace Status Ranking Review using the NatureServe/Heritage Program Methodology.
Dawley, R. M., and K. A. Goddard. 1988. Diploid‐triploid mosaics among unisexual hybrids of the minnows Phoxinus eos and Phoxinus neogaeus. Evolution 42(4):649–659.
Dawley, R. M., Schultz, R. J., and K. A. Goddard. 1987. Clonal reproduction and polyploidy in unisexual hybrids of Phoxinus eos and Phoxinus neogaeus (Pisces; Cyprinidae). Copeia 1987:275–283.
Doeringsfeld, M. R., Schlosser, I. J., Elder, J. F., and D. P. Evenson. 2004. Phenotypic consequences of genetic variation in a gynogenetic complex of Phoxinus eos‐neogaeus clonal fish (Pisces: Cyprinidae) inhabiting a heterogeneous environment. Evolution 58(6):1261–1273.
Felts, E. 2013. Ecology of glacial relict fishes in South Dakota’s Sandhills region. Master’s thesis. South Dakota State University, Brookings.
Goddard, K. A., and R. M. Dawley. 1990. Clonal inheritance of a diploid nuclear genome by a hybrid freshwater minnow (Phoxinus eos‐neogaeus, Pisces: Cyprinidae). Evolution 44(4):1052–1065.
Goddard, K., R. Dawley, T. Dowling, and J. Bogart. 1989. Origin and genetic relationships of diploid, triploid, and diploid-triploid mosaic biotypes in the Phoxinus eos-neogaeus unisexual complex. Pages 268–280 in R. M. Dawley and J. P. Bogart, editors. Evolution and ecology of unisexual vertebrates. New York State Museum, Albany.
Goddard, K. A., O. Megwinoff, L. L. Wessner, and F. Giaimo. 1998. Confirmation of gynogenesis in Phoxinus eos-neogaeus (Pisces: Cyprinidae). Journal of Heredity 89:151–157.
Lee, D., C. Gilbert, C. Hocutt, R. Jenkins, D. McAllister, and J. Stauffer, Jr. 1980. Atlas of North American freshwater fishes. North Carolina Museum of Natural History, Raleigh.
Mee, J. A., and L. Rowe. 2010. Distribution of Phoxinus eos, Phoxinus neogaeus, and their asexually-reproducing hybrids (Pisces: Cyprinidae) in Algonquin Provincial Park, Ontario. PLoS ONE 5(10):1–6.
Mee, J. A., and E. B. Taylor. 2012. The cybrid invasion: widespread postglacial dispersal by Phoxinus (Pisces: Cyprinidae) cytoplasmic hybrids. Canadian Journal of Zoology 90:577–584.
MTAFS 2011. Species of Concern Status Webpage: https://units.fisheries.org/montana/science/species-of-concern/species-status/northern-redbelly-x-finescale-dace/
New, J. G. 1962. Hybridization between two Cyprinids, Chrosomus eos and Chrosomus neogaeus. Copeia 1962:147–152.
Stasiak, R. H. 1972. The morphology and life history of the finescale dace, Pfrille neogaea, in Itasca State Park, Minnesota. PhD Dissertation, University of Minnesota, Minneapolis.
Stasiak, R. H. 2006. Northern Redbelly dace (Phoxinus eos): a technical conservation assessment. Report to the U.S. Forest Service, Rocky Mountain Region, Species Conservation Project, Golden, Colorado.
Stringer, A. L. 2018. Status of Northern Pearl Dace and chrosomid dace in prairie streams of Montana. Master’s thesis. Montana State University, Bozeman.
Schlosser, I. J., M. R. Doeringsfeld, J. F. Elder, and L. F. Arzayus. 1998. Niche relationships of clonal and sexual fish in a heterogeneous landscape. Ecology 79:953–968.
Whittier, T. R., D. B. Halliwell, and S. G. Paulsen. 1997. Cyprinid distributions in Northeast U.S.A. lakes: evidence of regional-scale minnow biodiversity losses. Canadian Journal of Fisheries and Aquatic Sciences 54:1593–1607.

